Soil pH is one of those gardening concepts that sounds technical but has completely practical consequences. You can feed your soil generously, water correctly, and plant at the right time — and still get yellowing leaves, stunted plants, and poor yields if the pH is outside the range where your plants can actually absorb the nutrients you’re giving them.
Most gardeners know pH matters but aren’t entirely sure why, or what to do when something is off. This soil pH guide covers all of it: what the numbers mean, how pH affects nutrient availability and soil biology, how to test your soil accurately, and exactly what to add to raise or lower pH when correction is needed.
What Soil pH Actually Means
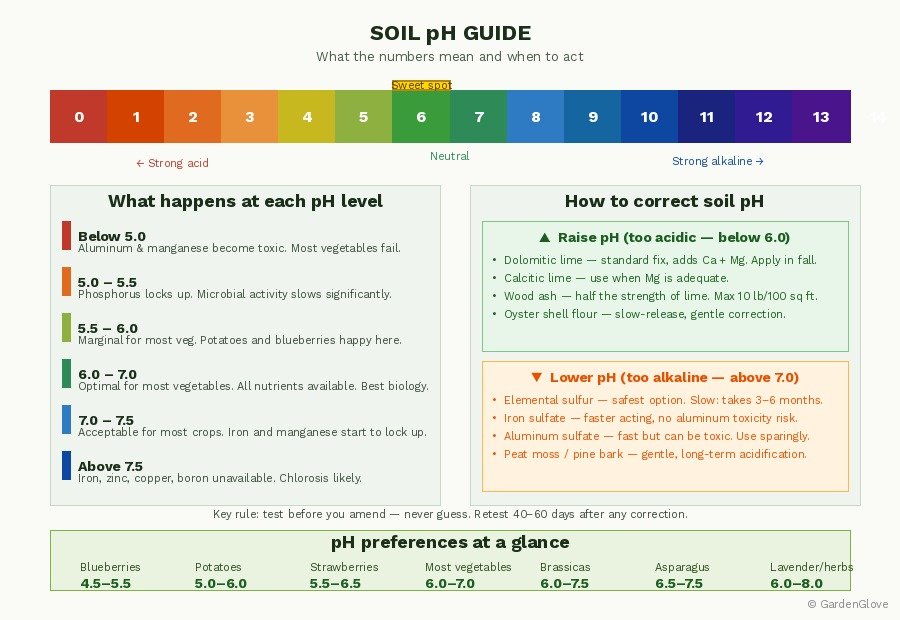
Soil pH is the measure of the concentration of hydrogen atoms in solution. The scale has a range from 0 to 14. Less than 7 is acidic, 7 is neutral, and greater than 7 is considered alkaline or basic.
The practical implication of the scale is something most people miss: the scale is not linear. Soil with a pH of 5.5 is 10 times more acidic than soil with a pH of 6.5 and 100 times more acidic than soil with a pH of 7.5. This means small numerical movements on the pH scale represent large chemical changes in the soil. A correction from 5.5 to 6.5 is a tenfold shift — not a small adjustment.
This also means overcorrection is a real risk. Gardeners who try to raise pH from 5.5 to 6.5 and slightly overshoot to 7.0 haven’t made a minor error — they’ve gone from mildly acidic to neutral, which can create a completely different set of nutrient availability problems. Don’t overcorrect. Make small changes and retest.
Why pH Matters — The Nutrient Availability Mechanism
The reason pH matters so profoundly in gardening isn’t chemistry for its own sake — it’s about whether plants can actually access the nutrients in the soil.
Beneath the soil surface, several chemical, biochemical, and biological reactions transform nutrients, whether naturally present or added through fertilizers and soil amendments, into forms plants can absorb. Soil pH controls many of these reactions by influencing the solubility of nutrients in soil water. The more soluble a nutrient is, the more readily it dissolves into the water around plant roots, allowing for easier uptake.
The practical consequence: a soil that tests adequate in nitrogen, phosphorus, and potassium can still produce nutrient-deficient plants if pH is significantly off. The nutrients are physically present but chemically inaccessible. You could fertilize heavily and see no improvement — or even see things get worse — because the fertilizer you’re adding is just joining the locked-up pile.
Each nutrient has its own optimal pH range for availability. In acidic soils (below 5.5), phosphorus, potassium, and calcium become tightly bound and inaccessible. The micronutrients iron, manganese, and zinc become more available as pH decreases, but molybdenum becomes less available. At very low pH (below 5.0), aluminum and manganese can actually reach toxic levels. Above 7.5, essential nutrients, including micronutrients like iron, copper, manganese, zinc, and boron, may again become inaccessible.
The sweet spot for the widest range of nutrient availability is 6.0–7.0. Nearly every vegetable species will experience optimal growth when the pH is approximately 6.5, and the rest will still be within tolerance.
How pH Affects Soil Biology
Nutrients aren’t the only thing pH controls. Soil biology — the bacteria, fungi, earthworms, and other organisms that make a productive garden possible — is also pH-sensitive.
In acidic soils with pH below 5.5, microbial activity slows, reducing the breakdown of organic matter. Bacteria are generally more prevalent at higher pH, while fungi are more prevalent at lower pH. A pH between 6 and 7 is ideal for maximizing both. This is important because microbes are responsible for making nutrients like nitrogen, phosphorus, and sulfur available to plants.
This adds another layer to the pH problem. At low pH, it’s not just that nutrients are chemically locked up — the biological processes that convert organic matter into plant-available nutrients slow down as well. Compost you add to an acidic bed breaks down more slowly and releases fewer nutrients than the same compost added to a bed at pH 6.5. Everything in the soil system works better in the right pH range.
What pH Range Your Plants Actually Need
Most vegetables share a similar preferred range, which makes managing a mixed kitchen garden relatively straightforward. Soil pH in the 6.0 to 7.0 range is optimal for the health of most vegetable plants.
Here’s a practical breakdown by crop category:
| Crop | Preferred pH Range |
|---|---|
| Most vegetables (tomatoes, peppers, beans, cucumbers, squash) | 6.0–7.0 |
| Brassicas (broccoli, cabbage, kale, cauliflower) | 6.0–7.5 |
| Root vegetables (carrots, beets, onions, parsnips) | 6.0–7.0 |
| Lettuce and salad greens | 6.0–7.0 |
| Potatoes | 4.8–6.0 (lower pH reduces scab disease) |
| Blueberries | 4.5–5.5 |
| Strawberries | 5.5–6.5 |
| Raspberries and blackberries | 5.5–6.5 |
| Roses | 6.0–7.0 |
| Rhododendrons, azaleas | 4.5–6.0 |
| Lavender, rosemary, thyme | 6.0–8.0 |
| Asparagus | 6.5–7.5 |
| Corn | 5.8–7.0 |
| Peas | 6.0–7.5 |
The practical takeaway for a mixed vegetable garden: aim for 6.0–6.8. This range keeps the majority of vegetables in their comfort zone and maintains optimal soil biological activity. Most gardeners opt for a compromise solution and look for a pH range of 6.0 to 7.0 in selecting a garden site.
The exceptions worth noting: potatoes prefer lower pH than most vegetables — scab-susceptible potato varieties are an exception to the standard 6.5–6.8 target, performing better and showing less disease in the 5.0–6.0 range. Blueberries require genuinely acidic conditions (4.5–5.5) and are difficult to grow successfully in a mixed vegetable bed — they’re better suited to a dedicated acidified planting area.
How to Test Your Soil pH
Never adjust soil pH without testing first. The most common soil pH mistake is amending based on assumption — adding lime because the soil “looks acidic” or because the previous owners complained about it. You can easily make a problem worse by correcting something that didn’t need correcting.
Home Test Kits
Home soil test kits are available at any garden center for $15–$25. They measure pH and the three major nutrients (N, P, K) quickly and without sending samples away. The results are adequate for a rough read — they’ll tell you whether you’re in the right ballpark or significantly off.
The limitation is precision. Home kits typically read in 0.5 pH increments, which can leave you uncertain whether you’re at 6.0 or 6.5. A pH test number that is more than 0.5 on either side of the optimal pH number for the plants you want to grow will require a soil amendment or additive to adjust the pH. A pH test number that is within 0.5 of the optimal pH number for the plants you want to grow does not require soil amendments.
Laboratory Soil Tests
A laboratory test from your state’s cooperative extension service provides significantly more precise readings — typically to 0.1 pH units — along with major and minor nutrient levels, organic matter percentage, and specific amendment recommendations tailored to your target crops. The best way to determine the lime requirement for a soil is to take a soil sample to your local extension soil testing laboratory. Costs run $15–$25 and results typically arrive within a week.
For a new garden, a garden with persistent plant health problems, or any garden you’re making significant investments in, the laboratory test is worth the extra step. The amendment recommendations it provides are specific enough to prevent the overcorrection that home kits can lead to.
How to collect a good soil sample: Take small amounts from 8–10 spots around the bed at 4–6 inch depth. Mix them together in a clean container. Allow to air dry if very wet. Send about a cup of this composite sample. A composite sample gives a representative reading of the whole bed rather than reflecting one unusual patch.
How to Raise Soil pH (Make It Less Acidic)
Acidic soil — pH below 6.0 for most vegetables — is the more common problem in the US, particularly in the Pacific Northwest, Southeast, and the Eastern Seaboard where rainfall is high and soils naturally acidify over time.
Agricultural Lime (Ground Limestone)
Overly acidic soil is neutralized with the addition of limestone. Powdered or pelleted agricultural limestone is most commonly used. Lime is the standard, reliable, and inexpensive fix for acidic soil. It works slowly — the reaction with soil takes weeks to months — and should ideally be incorporated before planting rather than applied to an established garden.
Two main types are available:
Calcitic lime (calcium carbonate): Standard agricultural lime. Raises pH and adds calcium. The right choice when soil calcium is adequate or when magnesium is not deficient.
Dolomitic lime (calcium magnesium carbonate): Raises pH while adding both calcium and magnesium. Dolomitic limestone, containing both elements, should be used when magnesium levels are low in soil. This is the more commonly recommended choice for vegetable gardens because magnesium deficiency is relatively common in acidic soils.
Application rate depends on soil texture — sandy soils require half-rate applications to achieve the desired pH change compared to clay or loam, because the buffering capacity of the soil (its resistance to pH change) varies with texture. Follow soil test rate recommendations, or start conservatively and retest.
Timing: It is best to mix liming material into the soil in the fall after harvest. This gives lime time to react through winter, and the pH will have stabilized by spring planting. For significant corrections, when a large amount of material is recommended, mix half in the fall and the other half in the spring before planting.
Wood Ash
Wood ash from untreated wood is a secondary option for raising pH. The calcium carbonate equivalence of wood ash is typically between 30–50% — meaning it takes roughly twice as much wood ash as lime to achieve the same pH change. It also adds potassium, making it useful as a combined pH and fertility amendment when potassium is needed.
Do not apply more than 10 lbs. of wood ashes per 100 square feet, and avoid applying both lime and wood ashes as these actions can push pH too high. Never use ash from treated wood, charcoal, or trash — only clean hardwood ash.
Oyster Shell Flour
Crushed oyster shells release calcium carbonate slowly, raising pH gradually over time. More expensive than agricultural lime but gentler — a good choice for incremental correction or for gardeners who want to avoid overcorrection.
How to Lower Soil pH (Make It More Acidic)
Alkaline soil — pH above 7.0 — is more common in arid western regions, in areas with limestone geology, and sometimes as the result of over-application of lime or wood ash in established gardens.
Elemental Sulfur
Elemental sulfur is the safest and most reliable amendment for lowering soil pH. Elemental sulfur must go through two processes, a biological process and a chemical process, before soil pH is decreased. This often takes 3 to 6 months of warm soil temperatures when soil biology is active. It’s slow but safe and inexpensive.
Apply in spring or early fall when soil is warm enough for biological activity. Sulfur-based materials that lower pH require heat to react with your soil, so are best applied in spring. Work into the top 6 inches before planting for best results.
To correct alkaline soil, add elemental sulfur at the rate of 4 pounds per 100 square feet for each unit of pH above 7. Follow soil test recommendations for your specific situation — application rates vary by soil texture.
Aluminum Sulfate
Aluminum sulfate reacts faster than elemental sulfur — the change in pH happens within days or weeks. However, aluminum sulfate is not an ideal amendment because it requires more material than elemental sulfur to reduce the soil pH, and aluminum is toxic to plants. Use sparingly and only when a faster correction is needed. It can accumulate to harmful levels if applied repeatedly.
Iron Sulfate
A faster-acting alternative to elemental sulfur without the aluminum toxicity concern. Iron sulfate is a go-to for correcting alkaline soils when quicker results are needed than elemental sulfur provides. Apply per soil test recommendations.
Acidic Organic Matter
For mild alkalinity (pH 7.0–7.5), acidic organic amendments can make a meaningful contribution over time. The best way to lower soil pH to neutral 6.0 to 7.0 is to add naturally acidic organic materials. These include peat moss, pine bark mulch, pine needle mulch, cottonseed meal, and coffee grounds (used sparingly). These options work slowly — expect improvements over multiple seasons rather than in a single application — but they improve soil structure simultaneously, making them the preferred approach when the correction needed is modest.
The Limits of pH Correction — What You Need to Know
Major pH corrections generally do not produce lasting results. This is one of the most important things to understand before investing heavily in pH amendment.
Soil pH is largely determined by the underlying geology and climate of the region. Rainfall leaches calcium and magnesium, gradually acidifying soil over time — which is why soils in high-rainfall regions are naturally acidic and require periodic re-liming. In alkaline regions, the carbonate minerals in the bedrock continually buffer soil toward high pH. You can make corrections, but without addressing the underlying chemistry, soils will tend to revert toward their natural state.
The practical implications:
Corrections need to be maintained. Acidic soils in high-rainfall areas will need lime reapplied every few years as pH drifts back down. Alkaline soils improved with sulfur may gradually revert, particularly in limestone regions.
Dramatic corrections are rarely sustainable. Trying to grow blueberries (pH 4.5–5.5) in naturally alkaline soil (pH 7.5+) by acidifying is a perpetual battle. In general, matching plant species to soil characteristics is easier, less expensive, and more reliable than adjusting soil pH. For highly pH-specific crops in incompatible soil, a raised bed filled with the right growing medium is often the most practical solution.
Compost moderates pH naturally. Add compost regularly. It naturally balances pH and improves structure. Consistent compost addition buffers soil pH, resisting movement in either direction. A well-amended, organic-matter-rich soil is more pH-stable than raw mineral soil and requires less frequent correction.
Test before you treat, and retest after. After adding sulfur or limestone or any soil additive to your garden, retest the soil in 40 to 60 days. Expect small changes to the pH — 0.5 to 1 unit at most. Testing every two to three seasons in a maintained garden is sufficient to catch drift before it becomes a problem.
Recognizing pH Problems in the Garden — What to Look For
You don’t always need a test to suspect a pH problem. Plants show recognizable symptoms when pH is significantly off.
Yellowing between leaf veins on young leaves (interveinal chlorosis): The most common sign of high pH (above 7.0). Iron, manganese, and zinc become unavailable in alkaline conditions — the yellow tissue between green veins is the visible result. Iron or zinc deficiencies in the garden are often due to above-optimum soil pH and can be corrected by reducing the soil pH rather than adding additional micronutrients. Adding iron fertilizer without correcting pH is a temporary fix at best.
Stunted plants, dark or bluish-green leaves: Often associated with phosphorus lockout at low pH. Aluminum toxicity (below pH 5.0) can produce similar symptoms.
Poor germination, rotting seedlings: Very acidic soil inhibits beneficial soil bacteria that protect seeds and seedlings from soil-borne pathogens. Germination rates drop noticeably below pH 5.5.
Plants that grow poorly despite apparent good care: If watering, fertilizing, and light seem adequate but plants chronically underperform, pH is worth testing. It’s the hidden variable that resists other interventions.
Purple coloration on leaves: Can indicate phosphorus deficiency from low pH locking it out, or cold soil — check pH and temperature together.
pH Testing Frequency
For a new garden bed: test before you plant and again after any significant amendment. For an established vegetable garden: testing the soil once every 3 years to monitor pH is recommended. For perennial plantings, fruit trees, or pH-sensitive crops like blueberries: test annually until stable, then every two years.
The investment — $15–$25 and 20 minutes — is the most efficient single step toward a productive garden. Every other practice in a well-managed garden depends on getting this variable right.
For related guides, see our Best Garden Soil Guide, Raised Bed Fertilizer Guide, and Clay Soil Improvement.

